CN
CN
 CN
CN
 CN
CN

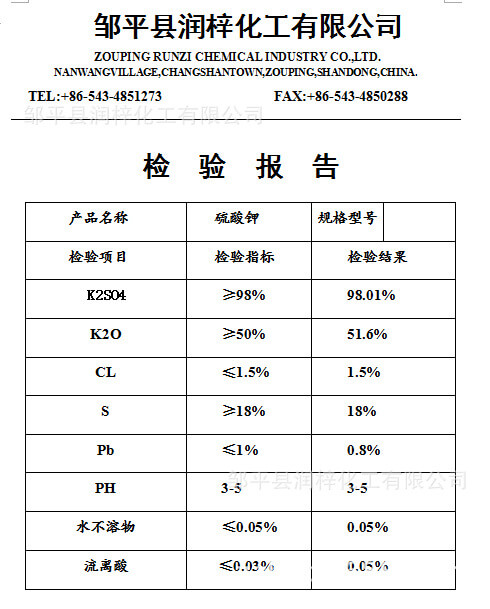
Potassium sulfate
Chemical formula: K2SO4
Density: 2.66 g/cm³, anhydrous
Melting point: 400°C
Solubility: 110 g/L (20°C), readily soluble in water.
Chemical properties: The aqueous solution is neutral.
Double displacement reaction: It can react with soluble barium salt solutions to form a barium sulfate precipitate.
Potassium sulfate is a colorless crystalline substance with low hygroscopicity, resistant to caking, and excellent physical properties, making it easy to apply. It serves as an excellent water-soluble potassium fertilizer. Additionally, potassium sulfate is chemically neutral but physiologically acidic.
Purpose: Used in the military industry as a smoke suppressant for artillery shells. It serves as a raw material for producing potassium salts. In the dye industry, it functions as an intermediate. In the glass industry, it acts as a clarifying agent. In medicine, it is employed as a mild laxative. In the fragrance industry, it serves as an additive. In the food industry, it functions as a general-purpose additive. It is also utilized in biochemical tests for serum proteins.
| 200-500 mesh boric acid (dissolves in water) | 200-500 mesh borax (soluble in water) |
| 200-500 mesh magnesium sulfate (soluble in water) | 200-500 mesh zinc sulfate (soluble in water) |
| 200-500 mesh copper sulfate (soluble in water) | 200-500 mesh ammonium sulfate (soluble in water) |
| 200-500 mesh manganese sulfate (soluble in water) | 200-500 mesh potassium sulfate (soluble in water) |
| 200-500 mesh ferrous sulfate (soluble in water) |